We’ve all been there, that nagging pain that creeps in during a run, threatening to derail your training. Running injuries…


We’ve all been there, that nagging pain that creeps in during a run, threatening to derail your training. Running injuries…
The Food and Drug Administration (FDA) stands as a cornerstone of the United States healthcare system, tasked with the monumental…

In the complex and highly regulated world of healthcare, the introduction of the Electronic Common Technical Document (eCTD) has revolutionized…
Liposuction’s journey from its inception to becoming one of the most sought-after cosmetic procedures is a fascinating tale of innovation,…

In the realm of cosmetic enhancement, liposuction emerges as a pivotal solution for individuals aiming to refine and sculpt their…
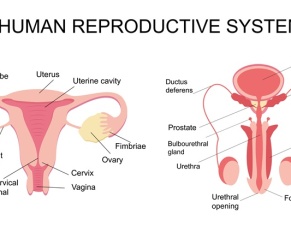
Male infertility, a complex and often misunderstood condition, affects a significant portion of the population. Understanding its causes and implications,…

Staying physically active is a cornerstone of healthy aging. As we grow older, our bodies and health needs change, making…
Understanding Facet Arthrosis: A Guide to Spinal Health Facet arthrosis, also known as facet joint arthritis, poses a significant challenge…
Lumbar laminectomy, a surgical procedure designed to alleviate back pain and discomfort caused by spinal stenosis, has emerged as a…

In today’s fast-paced world, back pain has emerged as a common ailment affecting individuals of all ages, such as the…